KTGHard
Question
A fixed container is fitted with a piston which is attached to a spring of spring constant k. The other end of the spring is attached to a rigid wall. Initially the spring is in its natural length and the length of container between the piston and its side wall is L. Now an ideal diatomic gas is slowly filled in the container so that the piston moves quasistatically. It pushed the piston by x so that the spring now is compressed by x. The total rotational kinetic energy of the gas molecules in terms of the displacement x of the piston is (there is vacuum outside the container)
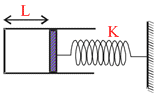

Options
A.k x L
B.4k x L
C.kx(x + L)
D.2kx2/ L
Solution
Rotational K.E. = Rotational degree of freedom x 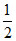 nRT
nRT
= 2 x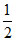 nRT = nRT = PV
nRT = nRT = PV
= PA.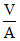 = force on piston × (L + x) = kx (L + x)
= force on piston × (L + x) = kx (L + x)
= 2 x
= PA.
Create a free account to view solution
View Solution FreeMore KTG Questions
Ideal gas is taken through process shown in figure:...Two different ideal diatomic gases A and B are initially in the same state. A and B are then expanded to same final volu...The expansion of an ideal gas of mass m in volume at a constant pressure P is given by the straight line B. Then the exp...What is critical temperature, If a and b are vander wall′s constant....The temperature of ideal gas is the result of which of the following energies :...