KTGHard
Question
Ideal gas is taken through process shown in figure:
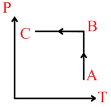

Options
A.In process AB, work done by system is positive
B.In process AB, heat is rejected out of the system.
C.In process AB, internal energy increases
D.In process AB internal energy decreases and in process BC internal energy increases.
Solution
In process AB T = constant P = increases P α 1/V
or V = decreases ᐃQ = ᐃW . ᐃW = − ve. or ᐃQ = − ve
∴ heat is rejected out of the system.
or V = decreases ᐃQ = ᐃW . ᐃW = − ve. or ᐃQ = − ve
∴ heat is rejected out of the system.
Create a free account to view solution
View Solution FreeMore KTG Questions
The constant quantity for equal volume of two gases at same pressure and temperature is-...In an adiabatic process on a gas with g = 1.4, the pressure is increased by 0.5%. The volume decreases by about...The presence of atmosphere on a planet means :- (Ves − escape velocity)...In the above question, minimum pressure attainable is...A cyclic process of an ideal monoatomic gas is shown in figure. The correct statement is (are) :...