KTGHard
Question
Two different ideal diatomic gases A and B are initially in the same state. A and B are then expanded to same final volume through adiabatic and isothermal process respectively. If PA , PB and TA , TB represents the final pressure and temperatures of A and B respectively then:
Options
A.PA < PB and TA < TB
B.PA > PB and TA > TB
C.PA > PB and TA < TB
D.PA < PB and TA > TB
Solution
(A)
For adiabatic process
P V1γ = PA V2γ
PA = P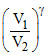 ......(1)
......(1)
For isothermal process
P V1 = PB V2
PB = P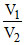 .....(2)
.....(2)
From (1) and (2)
PA < PB [For expansion V2 > V1]
and by PV = nRT TA < TB
For adiabatic process
P V1γ = PA V2γ
PA = P
For isothermal process
P V1 = PB V2
PB = P
From (1) and (2)
PA < PB [For expansion V2 > V1]
and by PV = nRT TA < TB
Create a free account to view solution
View Solution FreeMore KTG Questions
Which of the following is correct for the molecules of a gas in thermal equilibrium ?...The pressure P and volume V of an ideal gas both decreases in a process....Keeping the number of moles, volume and pressure the same, which of the following are the same for all ideal gas ?...For an ideal gas, the initial pressure and volume are equal to the final pressure and volume....A real gas behaves like an ideal gas if its...