KTGHard
Question
The expansion of an ideal gas of mass m in volume at a constant pressure P is given by the straight line B. Then the expansion of the same ideal gas of mass 2m at a pressure 2P is given by the straingth line.
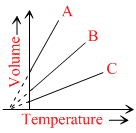

Options
A.C
B.A
C.B
D.none
More KTG Questions
In a cyclic process shown on the P − V diagram the magnitude of the work done is :...Hydrogen and helium volume V at same temperature T and same pressure P are mixed to have same volume V. The resulting pr...If the ratio of specific heats is γ and R is the gas constant then what will be the value of specific heat for gas ...The internal energy of an ideal gas is equal to negative of the work done by the system, then...According to kinetic theory of gases the relation between internal energy U and absolute temperature T is -...