KTGHard
Question
A diatomic ideal gas undergoes a thermodynamic change according to the P−V diagram shown in the figure. The total heat given to the gas is nearly (use ln2 = 0.7) :
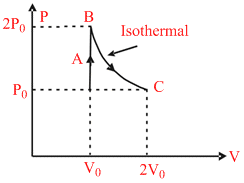

Options
A.2.5 P0V0
B.1.4 P0V0
C.3.9 P0V0
D.1.1 P0V0
Solution
QAB = ᐃ UAB + WAB
WAB = 0
ᐃ UAB =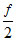 nR ᐃ T ⇒
nR ᐃ T ⇒ 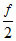 (ᐃ PV)
(ᐃ PV)
ᐃ UAB = (ᐃ PV)
QAB = 2.5 P0 V0
Process BC
QBC = ᐃ UBC + WBC
QBC = 0 + 2P0 V0 ln 2 = 1.4 P0 V0
Qnet = QAB + QBC = 3.9 P0 V0
WAB = 0
ᐃ UAB =
ᐃ UAB = (ᐃ PV)
QAB = 2.5 P0 V0
Process BC
QBC = ᐃ UBC + WBC
QBC = 0 + 2P0 V0 ln 2 = 1.4 P0 V0
Qnet = QAB + QBC = 3.9 P0 V0
Create a free account to view solution
View Solution FreeMore KTG Questions
In a cyclic process shown in the figure an ideal gas is adiabatically taken from B and A, the work done on the gas durin...When an ideal gas undergoes an adiabatic change causing a temperature change ᐃT(i) there is no heat gained or lost...The kinetic energy associated with per degree of freedom of a molecule is-...One mole of an ideal gas undergoes a process in which T = T0 + aV3, where T0 and ′a′ are positive constants ...A thermally insulated chamber of volume 2V0 is divided by a frictionless piston of area S & mass m into two equal parts ...