KTGHard
Question
One mole of an ideal gas undergoes a process in which T = T0 + aV3, where T0 and ′a′ are positive constants and V is volume. The volume for which pressure will be minimum is
Options
A.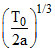
B.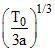
C.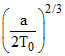
D.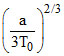
Solution
(A)
T = T0 + aV3
⇒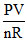 = T0 + aV3
= T0 + aV3
⇒ P = nR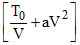
For minimum P,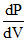 = O
= O
⇒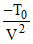 + a 2V = 0 ⇒ V =
+ a 2V = 0 ⇒ V = 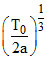
T = T0 + aV3
⇒
⇒ P = nR
For minimum P,
⇒
Create a free account to view solution
View Solution FreeMore KTG Questions
The pressure P and volume V of an ideal gas both decreases in a process....For an ideal gas, the initial pressure and volume are equal to the final pressure and volume....A fixed container is fitted with a piston which is attached to a spring of spring constant k. The other end of the sprin...The reason for the absence of atmosphere on moon is that the :...The ratio ofat a given temperature is :-...