KTGHard
Question
A thermally insulated chamber of volume 2V0 is divided by a frictionless piston of area S & mass m into two equal parts A and B.Part A has an ideal gas at pressure P0 and temperature T0 and in part B is vacuum. A massless spring of force constant K is connected with the piston and the wall of the container as shown. Initially the spring is undeformed. The gas in chamber A is allowed to expand. Let in equilibrium the spring is compressed by x0 . Then :
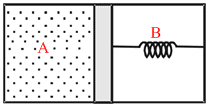

Options
A.Pressure of the gas at equilibrium is 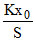
B.Work done by the gas is 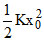
C.Increase in internal energy of the gas is 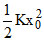
D.Increase in internal energy of the gas is
Solution
Temperature of the gas is decreased
(A) (D)
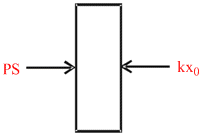
For equilibrium of piston
PS = Kx0
P =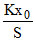
For piston
Wall = KE2 − KE1
Wgas −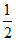 kx2 =
kx2 = 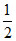 mv2
mv2
Wgas =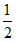 kx2 +
kx2 + 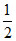 mv2 = position
mv2 = position
ᐃQ = 0
ᐃQ = ᐃU + W
ᐃU = − W = negative
As internal energy of gas decreases ∴ temperature of gas decreases.
(A) (D)

For equilibrium of piston
PS = Kx0
P =
For piston
Wall = KE2 − KE1
Wgas −
Wgas =
ᐃQ = 0
ᐃQ = ᐃU + W
ᐃU = − W = negative
As internal energy of gas decreases ∴ temperature of gas decreases.
Create a free account to view solution
View Solution FreeMore KTG Questions
Which of the following statement is not according to the postulates of kinetic theory of gases:-...Which gas has maximum root mean square speed at same temperature and pressure conditions-...P-T diagram is shown below then choose the corresponding V-T diagram...N(< 100) molecules of a gas have velocities 1, 2, 3........ N km/s respectively. Then...If K is Boltzman′s constant, m the mass of one molecule and temperature of gas is T, then root mean square velocit...