KTGHard
Question
In a cyclic process shown in the figure an ideal gas is adiabatically taken from B and A, the work done on the gas during the process B → A is 30 J, when the gas is taken from A → B the heat
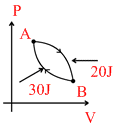

Options
A.20 J
B.− 30 J
C.50 J
D.− 10 J
Solution
B → A
ᐃQ = 0
0 = − 30 + ᐃU
ᐃUBA = 30 J
∴ ᐃUAB = −ᐃUBA = − 30 J
ᐃQ = 0
0 = − 30 + ᐃU
ᐃUBA = 30 J
∴ ᐃUAB = −ᐃUBA = − 30 J
Create a free account to view solution
View Solution FreeMore KTG Questions
The kinetic energy associated with per degree of freedom of a molecule is-...In vanderwall′s equation, (V − B) = RT, the dimension of a is :-...Pi, Vi are initial pressure and volumes and Vf is final volume of a gas in a thermodynamic process respectively. If PVn ...Cooking gas containers are kept in a lorry moving with uniform speed. The temperature of the gas molecules inside will...Three moles of an ideal monoatomic gas perform a cycle shown in figure. The gas temperatures in different states are T1 ...