KTGHard
Question
Given that the equilibrium constant for the reaction
2SO2(g) + O2(g) ⇋ 2SO3(g)
has a value of 278 at a particular temperature. What is the value of the equilibrium constant for the following reaction at the same temperature.
SO3(g) ⇋ SO2(g) + 1/2 O2(g)
2SO2(g) + O2(g) ⇋ 2SO3(g)
has a value of 278 at a particular temperature. What is the value of the equilibrium constant for the following reaction at the same temperature.
SO3(g) ⇋ SO2(g) + 1/2 O2(g)
Options
A.1.3 × 10-5
B.1.8 × 10-3
C.1.6 × 10-3
D.6.0 × 10-2
Solution
2SO2 + O2 ⇋ 2SO3
Keq = 278
SO3 ⇋ SO2 + 1/2 O2
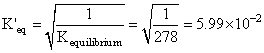
Keq = 278
SO3 ⇋ SO2 + 1/2 O2
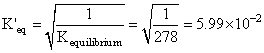
Create a free account to view solution
View Solution FreeMore KTG Questions
For an ideal gas, the initial pressure and volume are equal to the final pressure and volume....At what temperature will the rms velocity of O2 molecules be equal to that of H2 at 200K...The pressure of an ideal gas is written as E = . Here E stands for...In the above question, if the work done on the system along the curved path ′ba′ is 52J, heat abosrbed is...Starting with the same initial conditions, an ideal gas expands from volume V1 to V2 in three different ways. The work d...