KTGHard
Question
The pressure of an ideal gas is written as E = 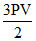 . Here E stands for
. Here E stands for
Options
A.total translational kinetic energy
B.rotational kinetic energy
C.average translational kinetic energy
D.total kinetic energy.
Solution
As translation K.E is K.E = 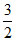 nRT E =
nRT E = 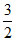 PV
PV
where E = total translational K.E.
where E = total translational K.E.
Create a free account to view solution
View Solution FreeMore KTG Questions
The r.m.s. speed of a gas molecule is 300 m/sec. Calculate the r.m.s. speed if the molecular weight is doubled while the...If K is Boltzman′s constant, m the mass of one molecule and temperature of gas is T, then root mean square velocit...A gas contained in a box of 0.1 m3 at atmospheric pressure is connected to another vessel of 0.09 m3. Consequent change ...For an ideal gas, the heat capacity at constant pressure is larger than than that at constant volume because...N2 molecules is 14 times heavier than a H2 molecule. At what temperature will the rms speed of H2 molecules be equal to ...