KTGHard
Question
In figure, A and B are two adiabatic curves for two different gases. Then A and B corresponds to :
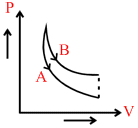

Options
A.Ar and He respectively
B.H2 and He respectivelyHe and H2 respectively
C.O2 and H2 respectively
D.H2 and He respectively
Solution
Slope = − 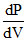
As slope of A > slope of B
∴ γ of A > γ of B
or A → Helium
B → Hydrogen
As slope of A > slope of B
∴ γ of A > γ of B
or A → Helium
B → Hydrogen
Create a free account to view solution
View Solution FreeMore KTG Questions
Suppose a container is evacuated to leave just one molecule of a gas in it. Let νmp and νav represent the most...The temperature of H2 and N2 are respectively 300 K and 450 K. The ratio of their average kinetic energies will be:-...The equation of state of a gas is given by VC = (RT + b), where a, b, c and R are constants. The isotherms can be repres...Consider a gas with density ρ and as the root mean square velocity of its molecules contained in a volume. If the s...The root mean square (rms) speed of oxygen molecules O2 at a certain temperature T (absolute) is υ. If the temperat...