KTGHard
Question
For a gas sample with N0 number of molecules, function N(V) is given by : N(V) = 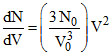 for 0 < V < V0 and N(V) = 0 for V > V0. Where dN is number of molecules in speed range V to V+ dV. The rms speed of the molecules is :
for 0 < V < V0 and N(V) = 0 for V > V0. Where dN is number of molecules in speed range V to V+ dV. The rms speed of the molecules is :
Options
A.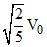
B.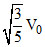
C.√2V0
D.√3V0
Solution
Vrms2 = < V2 > = 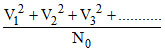
=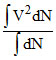 here
here 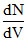 = N(V)
= N(V)
Vrms2 =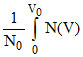 V2 dV =
V2 dV = 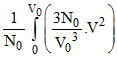 V2dV =
V2dV = 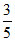 V02 ⇒ Vrms
V02 ⇒ Vrms 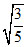 .V0.
.V0.
=
Vrms2 =
Create a free account to view solution
View Solution FreeMore KTG Questions
For an ideal gas, the heat capacity at constant pressure is larger than than that at constant volume because...A balloon contains 500 m3 of helium at 27o0C and 1 atmosphere pressure. The volume of the helium at - 3oC temperature an...At a given temperature, the pressure of an ideal gas of density ρ is proportional to -...The pressure of an ideal gas is written as E = . Here E stands for...28 gm of N2 gas is contained in a flask at a pressure 10 atm. and at a temperature of 57oC. It is found that due to leak...