KTGHard
Question
In a mixture of nitrogen and helium kept at room tempertaure. As compared to a helium molecule nitrogen molecule hits the wall
Options
A.With greater average speed
B.with smaller average speed
C.with greater average kinetic energy
D.with smaller average kinetic energy.
Solution
Vav α 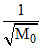
∴ oxygen molecule hits the wall with smaller average speed
∴ oxygen molecule hits the wall with smaller average speed
Create a free account to view solution
View Solution FreeMore KTG Questions
Which of the following statement is not true according to kinetic theory of gases :-...At what temperature will the rms speed of oxygen molecules become just sufficient for escaping from the Earth's atmo...Starting with the same initial conditions, an ideal gas expands from volume V1 to V2 in three different ways. The work d...The vander -Waal′s equation at :-...At 27o C temperature, the kinetic energy of an ideal gas is E1. If the temperature is increased to 327o C, then kinetic ...