KTGHard
Question
One mole of an ideal gas at a temperature T1K expands slowly according to the law = 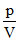 constant. Its final temperature is T2K. The work done by the gas is
constant. Its final temperature is T2K. The work done by the gas is
Options
A.R(T2 - T1)
B.2R(T2 - T1)
C.2R/3 (T2 - T1)
D.2R/3 (T2 - T1)
Solution
PV-1 = C
Comparing PVx = C
x = − 1
W =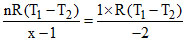
W =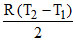
Comparing PVx = C
x = − 1
W =
W =
Create a free account to view solution
View Solution FreeMore KTG Questions
A thermodynamic process of one mole ideal mono atomic gas is shown in figure. The efficiency of cyclic process ABCA will...What is the meaning of point masses for the molecules of ideal gas :-...The temperature of ideal gas is the result of which of the following energies :...The root mean square and most probable speed of the molecules in a gas are :...Two samples 1 and 2 are initially kept in the same state. The sample 1 is expanded through an isothermal process where a...