KTGHard
Question
A one dimensional gas is a hypothetical gas with molecules that can move along only a single axis. The following table gives four situations, the velocities in meter per second of such a gas having four molecules. The plus and minus sign refer to the direction of the velocity along the axis.
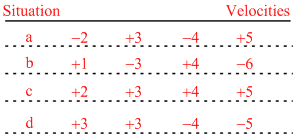
In which situation root-mean-square speed of the molecules is greatest

In which situation root-mean-square speed of the molecules is greatest
Options
A.a
B.b
C.c
D.d
Solution
Using Vrms = 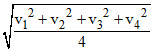
and substituting the corresponding velocity we get Vrms is greatest in situation b.
and substituting the corresponding velocity we get Vrms is greatest in situation b.
Create a free account to view solution
View Solution FreeMore KTG Questions
Equal volume of H2, O2 and He gases are at same temperature and pressure. Which of these will have large number of molec...The root mean square and most probable speed of the molecules in a gas are :...The root mean square speed of hydrogen molecules of an ideal hydrogen gas kept in a gas chamber at 0oC is 3180 m/s. The ...In given figure, a fixed mass of an ideal gas undergoes the change represented by XYZX below. Which one of the following...The lowest pressure (the best vaccum) that can be created in laboratory at 27oC is 10-11 mm of Hg. At this pressure, the...