KTGHard
Question
In the following V-T diagram what is the relation between P1 and P2 :
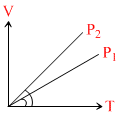

Options
A.P2 = P1
B.P2 > P1
C.P2 < P1
D.cannot be predicted
More KTG Questions
The rms velocity of gas molecules is C at temperature T. If the rms velocity is increased to 4 C, then what will be the ...Which of the following is correct for the molecules of a gas in thermal equilibrium ?...Consider a collision between an argon molecule and a nitrogen molecule in a mixture of argon and nitrogen kept at room t...Four containers are filled with mono atomic ideal gases. For each container, the number of moles, the mass of an individ...Figure shows a conducting cylinder containing gas and closed by a movable piston. The cylinder is submerged in an ice-wa...