NEETKTGHard
Question
Four containers are filled with mono atomic ideal gases. For each container, the number of moles, the mass of an individual atom and the rms speed of the atoms are expressed in terms of n, m and vrms respectively. If TA, TB, TC and TD are their temperatures respectively then which one of the options correctly represents the order ?
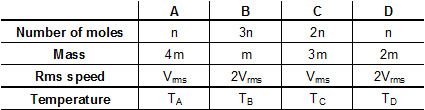
Options
A.TB = TC > TA > TD
B.TD > TA > TC > TB
C.TD > TA = TB > TC
D.TB > TC > TA > TD
Solution
Vrms = 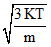 ⇒ T =
⇒ T = 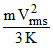
Therefore T ∝ mVrms2
hence option (c) is correct.
Therefore T ∝ mVrms2
hence option (c) is correct.
Create a free account to view solution
View Solution FreeMore KTG Questions
The mean kinetic energy of a molecule of an ideal gas is :...A student performs an experiment to determine the Young′s modulus of a wire, exactly 2 m long, by Searle′s m...A cylindrical tube $AB$ of length $l$, closed at both ends contains an ideal gas of 1 mol having molecular weight $M$. T...When an ideal diatomic gas is heated at constant pressure, the fraction of the heat energy supplied which increases the ...An ideal gas at 1 atmospheric pressure and at 273 K has 22.4 litre of volume. This is heated to 546 K and then by applyi...