KTGHard
Question
Which of the following is correct for the molecules of a gas in thermal equilibrium ?
Options
A.All have the same speed
B.All have different speeds which remain constant
C.They have a certain constant average speed
D.They do not collide with one another.
Solution
Vav = 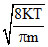 , as T = constant ∴ Vav = constant
, as T = constant ∴ Vav = constant
Create a free account to view solution
View Solution FreeMore KTG Questions
The temperature of H2 and N2 are respectively 300 K and 450 K. The ratio of their average kinetic energies will be:-...Let P1 and P2 be the final pressure of the samples 1 and 2 respectively in the previous question then :...If molar heat capacity of the given process (as shown in figure) is C, then...A one dimensional gas is a hypothetical gas with molecules that can move along only a single axis. The following table g...The critical temperature of CO2 is -...