KTGHard
Question
Figure shows a conducting cylinder containing gas and closed by a movable piston. The cylinder is submerged in an ice-water mixture. The piston is quickly pushed down from position (1) to position (2). The piston is held at position (2) until the gas is again at 0oC and then is slowly raised back to position (1).
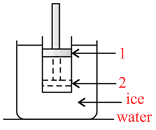
P-V diagram for the above process will be

P-V diagram for the above process will be
Options
A.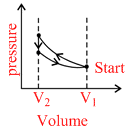

B.

C.

D.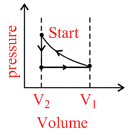

Solution
(A)
Correct graph is shown in option (A)
Process 1−2 adiabatic process, Process 2−3 Isochoric process, process 3−1 Isothermal process.
Correct graph is shown in option (A)
Process 1−2 adiabatic process, Process 2−3 Isochoric process, process 3−1 Isothermal process.
Create a free account to view solution
View Solution FreeMore KTG Questions
A gas contained in a box of 0.1 m3 at atmospheric pressure is connected to another vessel of 0.09 m3. Consequent change ...An air bubble of volume $2.9{\text{ }cm}^{3}$ rises from the bottom of a swimming pool of 5 m deep. At the bottom of the...The equation of state of a gas is given by VC = (RT + b), where a, b, c and R are constants. The isotherms can be repres...A mass of an ideal gas undergoes a reversible isothermal compression. Its molecules will then have compared with initial...Which of the following statement is not according to the postulates of kinetic theory of gases:-...