Ionic EquilibriumHard
Question
pH of a saturated solution of Ba ( OH )2 is 12. The value of solubility product Ksp of Ba ( OH2 ) is
Options
A.3.3 × 10-7
B.5.0 × 10-7
C.4.0 × 10-6
D.5.0 × 10-6
Solution
Given, pH of Ba ( OH )2 = 12
∴ pOH = 14 - pH
= 14 - 12 = 2
We know that,
pOH = - log [ OH- ]
2 = - log [ OH- ]
[ OH- ] = antilog ( -2 )
[ OH- ] = 1 × 10-2
Ba ( OH )2 dissolves in water as
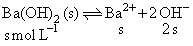
∴ [ OH- ] = 2s = 1 × 10 -2
[ Ba2+ ]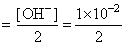
Ksp = [ Ba2+ ] [ OH- ]2
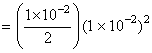
= 0.5 × 10-6 = 5 × 10-7
∴ pOH = 14 - pH
= 14 - 12 = 2
We know that,
pOH = - log [ OH- ]
2 = - log [ OH- ]
[ OH- ] = antilog ( -2 )
[ OH- ] = 1 × 10-2
Ba ( OH )2 dissolves in water as
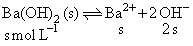
∴ [ OH- ] = 2s = 1 × 10 -2
[ Ba2+ ]
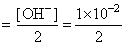
Ksp = [ Ba2+ ] [ OH- ]2
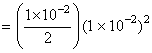
= 0.5 × 10-6 = 5 × 10-7
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
For the identification of β - naphthol using dye test, it is necessary to use...A volume of 2.5 ml of 2/5 M weak monoacidic base (Kb = 1 × 10−12 at 25o C) is titrated with (2/15) M-HCl in water at 25o...Which of the following solution will have pH close to 1.0 ?...At 90oC, pure water has [H3O+] as 10-6 mol -1. What is the value of Kw at 90oC ?...Solubility product of a salt AB is 1 × 10-8 in a solution in which th concentration of A+ ions is 10-3 M. The salt ...