Ionic EquilibriumHard
Question
Solubility product of a salt AB is 1 × 10-8 in a solution in which th concentration of A+ ions is 10-3 M. The salt will precipitate when the concentration of B- ions is kept
Options
A.between 10-8 M to 10-7 M
B.between 10-7 M to 10-8 M
C.> 10-5 M
D.< 10-8 M
Solution
AB ⇋ A+ + B-
Ksp - [A+][B-]
Salt will precipiteate of ionic conc. > Ksp
[A+][B-] > 1 × 10-8
(1 × 10-3)[B-] > 1 × 10-8
[B-] >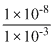 or 1 × 10-5
or 1 × 10-5
Ksp - [A+][B-]
Salt will precipiteate of ionic conc. > Ksp
[A+][B-] > 1 × 10-8
(1 × 10-3)[B-] > 1 × 10-8
[B-] >
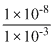 or 1 × 10-5
or 1 × 10-5Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
When weak base solution (50 mL of 0.1 N NH4OH) is titrated with strong acid (0.1 N HCl), the pH of the solution initiall...What is the pH of a neutral solution at 37°C, where Kw equals 2.5 × 10–14? (log 2 = 0.3)...In which of the following combinations, is buffer action expected?1. NH3 + NH4CI in 1 : 1 mole ratio2. HCI + NaCI in 1 :...Assuming no change in volume, calculate the minimum mass of NaCl necessary to dissolve 0.01 mole of AgCl in 100 L soluti...Which of the following is the strongest base :-...