Ionic EquilibriumHard
Question
Which of the following solution will have pH close to 1.0 ?
Options
A.00 mL of (M/ 10) HCl + 100 mL of (M/10)NaOH
B.55 mL of (M/ 10) HCl + 45 mL of (M/10)NaOH
C.10 mL of (M/ 10) HCl + 90 mL of (M/10)NaOH
D.75 mL of (M/ 5) HCl + 25 mL of (M/5)NaOH
Solution
HCl + NaOH → NaCl + H2O
Mwq.at t = 0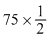
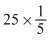
= 15 = 5 0 0
Meq. after reaction = 10 0 5 5
∴ NHCl =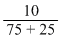 = 0.1 or 10-1
= 0.1 or 10-1
or [H+] = 10-1 ⇒ pH = - log 10-1 = 1
Mwq.at t = 0
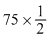
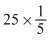
= 15 = 5 0 0
Meq. after reaction = 10 0 5 5
∴ NHCl =
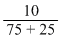 = 0.1 or 10-1
= 0.1 or 10-1 or [H+] = 10-1 ⇒ pH = - log 10-1 = 1
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
For a sparingly soluble salt ApBq the relationship of its solubility product (Ls) with its solubility (S) is :...An acid with molelcular formula C7H6O3 forms three types of sodium salts. i.e., C7H5O3Na, C7H4O3Na2 and C7H3O3Na3. The b...How many times the solubility of CaF2 is decreased in 4 × 10−3 M-KF(aq) solution as compared to pure water at 25o C. Giv...What mass of NaOH should be dissolved in sufficient water to get 20 m3 of an aqueous solution of pH 7.3 at 25o C?...Which of the following is the strongest base :-...