Gaseous StateHard
Question
Critical constant for a real gas are given as
TC = 180 K ; Vc = 0.1231 L/mole
PC = 45 atm ;
The correct statement for the real gas is -
TC = 180 K ; Vc = 0.1231 L/mole
PC = 45 atm ;
The correct statement for the real gas is -
Options
A.The actual volume of single gas molecule is 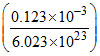
B.b = 3 × 0.123 L mol-1
C.The Boyle′s temperature is less than 180K
D.Gas cannot be liquified at 200K
Solution
(A) VC = 3 × b
= 3 × 4 × NA × volume of single molecule
volume of single molecule =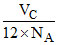
(B) b =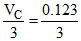
(C) TB =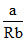 ⇒ TB > TC
⇒ TB > TC
(D) Above TC (Critical tempurature) gas cannot be liquified
= 3 × 4 × NA × volume of single molecule
volume of single molecule =
(B) b =
(C) TB =
(D) Above TC (Critical tempurature) gas cannot be liquified
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Assuming that O2 molecule is spherical in shape with radius 2(Ao), the percentage of the volume of O2 molecules to the t...For gaseous state, if most probable speed is denoted by C*, average speed by and mean square speed by C, then for a larg...A real gas most closely approaches the behaviour of an ideal gas at -...One litre of a gaseous mixture of two gases effuses in 311 seconds while 2 litres of oxygen takes 20 minutes. The vapour...The average kinetic energy of an ideal gas per molecule in S.I. units at 25oC will be ?...