Gaseous StateHard
Question
For the diffusion of a gas at pressure P, the rate of diffusion is expressed by:
Options
A.r ∝ 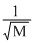
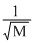
B.r = 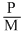
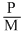
C.r ∝ 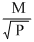
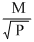
D.r = 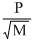
Solution
According to Graham′s law of diffusion or effusion ″under simillar conditions of temperature and pressure, rate of diffusion is inversely proportional to square root of molecular weight″ rα 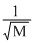
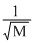
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
Van der waal′s equation (V - b) = nRT applicable for :...At Boyle′s temperature, the value of compressibility factor Z = (PVm / RT = Vreal/Videal) has a value of 1, over a...SO2 at STP contained in a flask was replaced by O2 under identical conditions of pressure, temperature and volume. Then ...Which of the following is/are correct for an mono atomic ideal gas molecules :...When a gas is compressed at constant temperature :...