Gaseous StateHard
Question
A mixture of hydrogen and oxygen at one bar pressure contains 20% by weight of hydrogen. Partial pressure of hydrogen will be
Options
A.0.2 bar
B.0.4 bar
C.0.6 bar
D.0.8 bar
Solution
(D) Weight of H2 = 20 g in 100 g mixture ; Weight of O2 = 80 g
∴ Moles of H2 =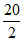 = 10 ; ∴ Moles of O2 =
= 10 ; ∴ Moles of O2 = 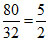
∴ Total moles = 10 +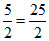
∴ PH2 = PT x mole fraction of H2 = 1 ×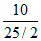 = 0.8 bar
= 0.8 bar
∴ Moles of H2 =
∴ Total moles = 10 +
∴ PH2 = PT x mole fraction of H2 = 1 ×
Create a free account to view solution
View Solution FreeMore Gaseous State Questions
If two moles of an ideal gas at 546 K occupies a volume of 44.8 litres, the pressure must be -...A thin balloon filled with air at 47oC has a volume of 3 litre. If on placing it in a cooled room its volume becomes 2.7...At 298 K, equal volumes SO2,CH4 and O2 are mixed in empty container. The total pressure of CH4 in mixture is :...0.28 g of a gas occupies 224 ml at STP. The gas could be...A gas described by van der Waal′s equation...