Ionic EquilibriumHard
Question
1.4 g of an organic compound was digested according to Kjeldahl′s method and the ammonia evolved was absorbed in 60 mL of M/10 H2SO4 solution. The excess sulphuric acid required 20 mL of M/10 NaOH solution for neutralization. The percentage of nitrogen in the compound is :
Options
A.24
B.5
C.10
D.3
Solution
Organic compound → NH3
(1.4g)
2NH3 + H2SO4 → (NH4)2SO4
H2SO4 + 2NaOH → Na2SO4 + 2H2O
nNH3 + 20 ×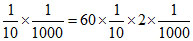
nNH3 =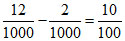
nN = nNH3 = 0.01 ⇒ mN = 0.01 × 14 = 0.14 ⇒ % of N =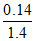 × 100 = 10%
× 100 = 10%
(1.4g)
2NH3 + H2SO4 → (NH4)2SO4
H2SO4 + 2NaOH → Na2SO4 + 2H2O
nNH3 + 20 ×
nNH3 =
nN = nNH3 = 0.01 ⇒ mN = 0.01 × 14 = 0.14 ⇒ % of N =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
Solubility product constant (Ksp) of salts of types MX, MX2 and M3X at temperature, T are 4.0 × 10−8, 3.2 × 10−14 and 2....The pH of 0.1 M solution of the following salts increases in the order:...Buffer solutions have constant acidity and alkalinity because...What will be the pH at the equivalence point during the titration of a 100 mL 0.2 M solution of CH3COONa with 0.2 M solu...Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term:...