Ionic EquilibriumHard
Question
What will be the pH at the equivalence point during the titration of a 100 mL 0.2 M solution of CH3COONa with 0.2 M solution of HCl ? Ka = 2 × 10-5.
Options
A.3 - log√2
B.3 + log√2
C.3 - log 2
D.3 + log 2
Solution
CH3COONa + HCI → NaCI + CH3COOH
t=0 20 m eq. 20 meq.
teq. - - 20 meq.
[CH3COOH] =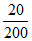 = 0.1 M
= 0.1 M
pH =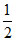 [pKa - log C] =
[pKa - log C] = 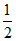 [5 - log2 + 1] =
[5 - log2 + 1] = 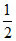 [6 - log2] = 3 - log√2
[6 - log2] = 3 - log√2
t=0 20 m eq. 20 meq.
teq. - - 20 meq.
[CH3COOH] =
pH =
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
pH of a saturated solution of Ba(OH)2 is 12. The value of solubility product (Ksp) of Ba(OH)2 is...At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At what pH, will Mg2+ ion start precipitating in the for...Which of the following is the strongest base :-...Which may be added to one litre of water to act a buffer:...Fear or excitement, generally cause one to breathe rapidly and it results in the decrease of concentration of CO2 in blo...