Ionic EquilibriumHard
Question
Solubility of BaF2 in a solution of Ba(NO3)2 will be represented by the concentration term:
Options
A.[Ba2+]
B.[F-]
C.1/2[F-]
D.2[NO3-]
Solution
Let S is the solubility of BaF2 in a solution of BaNO3
Then KSP = [Ba2+] [F-]2.
Then [F-] = 2S;
Then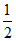 [F-] = S
[F-] = S
Then KSP = [Ba2+] [F-]2.
Then [F-] = 2S;
Then
Create a free account to view solution
View Solution FreeMore Ionic Equilibrium Questions
At 25oC, the solubility product of Mg(OH)2 is 1.0 × 10-11. At what pH, will Mg2+ ion start precipitating in the for...When 20 gm of naphthoic acid (C11H8O2) is dissolved in 50 gm of benzene (Kf = 1.72 Kg/m) a freezing point depression of ...When weak base solution (50 mL of 0.1 N NH4OH) is titrated with strong acid (0.1 N HCl), the pH of the solution initiall...The ionic product of water is 1.0 × 10−14 at 25o C. Assuming the density of water independent from change in temperature...Which of following can act as buffer ?...