Atomic StructureHard
Question
A sample of a hydrate of barium chloride weighing 61 g was heated until all the water of hydration is removed. The dried sample weighed 52g. The formula of the hydrated salt is: (atomic mass, Ba = 137 amu, Cl = 35.5 amu)
Options
A.BaCl2 + H2O
B.BaCl2 + 4H2O
C.BaCl2 + 3H2O
D.BaCl2 + 2H2O
Solution
BaCl2.xH2O → BaCl2 + xH2O.
mH2O = 61 - 52 = 9g
⇒ nH2O =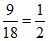
mBaCl2 = 52 ⇒ nBaCl2 =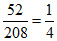
⇒ simplest formula =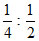 = 1 : 2 ⇒ BaCl2.2H2O
= 1 : 2 ⇒ BaCl2.2H2O
mH2O = 61 - 52 = 9g
⇒ nH2O =
mBaCl2 = 52 ⇒ nBaCl2 =
⇒ simplest formula =
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
In the structure of ClF3, the number of lone pairs of electrons on central atom 'Cl' is...If the value of principal quantum number is 4, the possible values for magnetic quantum number will be :-...The radii of two of the first four Bohr′s orbits of the hydrogen atom are in the ratio 1 : 4 The energy difference...If the shortest wave length of Lyman series of H atom is x, then the wave length of the first line of Balmer series of H...The electronic structure of zinc (Z = 30) is 2, 8, 18, 2. The electronic structure of gallium (Z = 31) will be...