Atomic StructureHard
Question
If the shortest wave length of Lyman series of H atom is x, then the wave length of the first line of Balmer series of H atom will be -
Options
A.9x/5
B.36x/5
C.5x/9
D.5x/36
Solution
Shortest wave length of Lyman series of H-atom
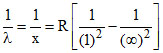 so, x =
so, x = 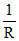
For Balmes series
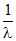 = R(1)2
= R(1)2 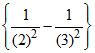
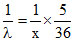 so, λ =
so, λ = 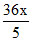
For Balmes series
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
O2 undergoes photochemical dissociation into one normal oxygen atom and one oxygen atom, 1.2 eV more energetic than norm...When alpha particles are sent through a thin metal foil, most of them go straight through the foil because :...Assume that the uncertainty in the position of a particle is equal to its de Broglie wavelength. The minimum uncertainly...If there are only two H-atoms, each is in 3rd excited state then :...For an electron in a hydrogen atom, the wave function is given by $\Psi_{1s} = \left( \pi/\sqrt{2} \right)e^{- r/a_{0}}$...