NEET | 2018Atomic StructureHard
Question
In the structure of ClF3, the number of lone pairs of electrons on central atom 'Cl' is
Options
A.
one
B.
two
C.
four
D.
three
Solution
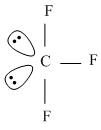
2 lone pair at equitorial position.
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
Which is true about an electron ?...The magnitude of spin angular momentum of an electron is given by...The de-Broglie wavelength of an electron in the ground state of hydrogen atom is :[K.E. = 13.6eV; leV = 602 × 10-19...The momentum of a photon of wavelength 6626 nm will be...The mass of a particle is 10–10 g and its diameter is 10−4 cm. If its speed is 10–6 cm/s with 0.0001% uncertainty in mea...