AIPMT | 2015Mole ConceptHard
Question
The Ksp of Ag2CrO4, AgCl, AgBr and AgI are respectively, 1.1 × 10-12, 1.8 × 10-10,. 5.0 × 10-13, 8.3 × 10-17. Which one of the following salts will precipitate last if AgNO3 solution is added to the solution containing equal moles of NaCl, NaBr, NaI and Na2CrO4 ?
Options
A.AgCl
B.AgBr
C.Ag2CrO4
D.AgI
Solution
Ag2CrO4 → KSP = [Ag+]2 [CrO4-2]
⇒ [Ag+] =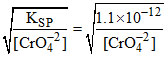 = maxm
= maxm
so answer is Ag2CrO4
⇒ [Ag+] =
so answer is Ag2CrO4
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Calculate the normality of 10 volume H2O2 ?...When the hydrocarbon propane is burned in air, carbon dioxide and water are formed. If 0.15 mol of CO2 is produced, then...A volume of 10 ml chlorine gas combines with 25 ml of oxygen gas to form 10 ml of a gaseous compound. If all the volumes...If the mass of neutron is doubled and that of proton is halved, the molecular mass of H2O containing only H1 and O16 ato...An organic compound contains C, H, and O. If C(%) : H(%) = 6 : 1, what is the simplest formula of the compound, given th...