Mole ConceptHard
Question
An organic compound contains C, H, and O. If C(%) : H(%) = 6 : 1, what is the simplest formula of the compound, given that one mole of the compound contains half as much oxygen as would be required to burn all the C and H atoms in it to CO2 and H2O ?
Options
A.CH2O
B.C2H4O3
C.C3H6O
D.C3H6O2
Solution
no. of moles= 4.2/420 =0.1
CxH2xOy + 1.5xO2 → xCO2 + xH2O
From the question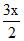 = y
= y
x : y = 2 : 3
Formula: C2H4O3.
no. of valence electron in 1 atom = 24
so ans is 0.1*24*NAMolar ratio of C:H = 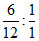 = 2:1
= 2:1
Formula of compound, CxH2xOy CxH2xOy + 1.5xO2 → xCO2 + xH2O
From the question
x : y = 2 : 3
Formula: C2H4O3.
Create a free account to view solution
View Solution FreeMore Mole Concept Questions
One mole of the aquous solution of complex CoCl3.4NH3 passess through anion exchange resine and resulting solution treat...Dissolving 120 g of urea (mol. wt. 60) in 1000 g of water gave a solution of density 1.15 g/mL. The molarity of the solu...In which of the following reactions, increase in the volume at constant temperature does not affect the number of moles ...Which of the following statements is/are correct ?20.0 mL of 6.0 M HCl is mixed with 50.0 mL of 2.0 M Ba(OH)2, and 30 mL...pH of a saturated solution of Ba ( OH )2 is 12. The value of solubility product Ksp of Ba ( OH2 ) is...