Mole ConceptHard
Question
Calculate the normality of 10 volume H2O2 ?
Options
A.1.7 N
B.12 N
C.30.3 N
D.0.0303 N
Solution
Normality of 10V of H2O2
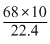 = 17 × N ∴ N = 1.78
= 17 × N ∴ N = 1.78
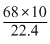 = 17 × N ∴ N = 1.78
= 17 × N ∴ N = 1.78Create a free account to view solution
View Solution FreeMore Mole Concept Questions
Silver metal in ore is dissolved by potassium cyanide solution in the presence of air by the reaction 4 Ag + 8 KCN + O2 ...Consider the following statements1. If all the reactants are not taken in their stoichiometric ratio, then at least one ...In which of the following pairs, 10 g of each have an equal number of molecules ?...In the reaction, 4A + 2B + 3C → A4B2C3, whatwill be the number of moles of product formed, starting from 1 mol of ...In the reaction2Al(s) + 6HCl(aq) → 2Al3+(aq) + 6Cl-(aq) + 3H2(g) :-...