Chemical Kinetics and Nuclear ChemistryHard
Question
For a first order reaction A → P, the temperature (T) dependent rate constant (k) was found to follow the equation log k = - (2000)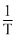 + 60. The pre-exponential factor A and the activation energy Ea, respectively, are
+ 60. The pre-exponential factor A and the activation energy Ea, respectively, are
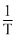 + 60. The pre-exponential factor A and the activation energy Ea, respectively, are
+ 60. The pre-exponential factor A and the activation energy Ea, respectively, areOptions
A.1.0 × 106 s-1 and 9.2 kJmol-1
B.6.0 s-1 and 16.6 kJmol-1
C.1.0 × 106 s-1 and 16.6 kJmol-1
D.1.0 × 106 s-1 and 38.3 kJmol-1
Solution
Given, log K = 6 - 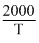
Since, log K = log A -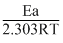 So, A = 106 sec-1 and Ea = 38.3 kJ/mole
So, A = 106 sec-1 and Ea = 38.3 kJ/mole
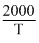
Since, log K = log A -
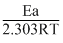 So, A = 106 sec-1 and Ea = 38.3 kJ/mole
So, A = 106 sec-1 and Ea = 38.3 kJ/moleCreate a free account to view solution
View Solution FreeTopic: Chemical Kinetics and Nuclear Chemistry·Practice all Chemical Kinetics and Nuclear Chemistry questions
More Chemical Kinetics and Nuclear Chemistry Questions
According to the Arrhenius equation, straight line is to be obtained by plotting the logarithm of the rate constant of a...In gaseous reactions important for the understanding of the upper atmosphere, H2O and O react bimolecularly to form two ...The rate constant is given by the equation $K = P.A.e^{- E_{a}/RT}$. Which factor should register a decrease for the rea...Higher order (>3) reactions are rare due to : -...The energies of activation for forward and reverse reactions for A2 + B2 ⇋ 2AB are 180 kJ mol-1 2AB are 180 kJ mol...