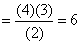Atomic StructureHard
Question
Hydrogen atom is excited from ground state toanother state with principal quantum numberequal to 4. Then the number of spectral lines in the emission spectra will be :-
Options
A.6
B.2
C.3
D.5
More Atomic Structure Questions
Which of the following has the highest neutrons ratio ?...A parallel beam of electrons travelling in x-direction falls on a slit of width d (see figure). If after passing the sli...Highly energetic electrons are bombarded on a target of an element containing 30 neutrons. The ratio of radii of nucleus...Mark the CORRECT option(s) : -...A radioactive material decays by simultaneous emission of two particles with respective half lives 1620 and 810 years. T...