Atomic StructureHard
Question
Highly energetic electrons are bombarded on a target of an element containing 30 neutrons. The ratio of radii of nucleus to that of Helium nucelus is 141/3. The atomic number of nucleus will be :-
Options
A.25
B.26
C.56
D.30
Solution
r ∝ A1/3
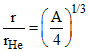 = (14)1/3 ⇒ A = 56
= (14)1/3 ⇒ A = 56
A = Z + N = 56 ⇒ Z = 56 - 30 = 26
A = Z + N = 56 ⇒ Z = 56 - 30 = 26
Create a free account to view solution
View Solution FreeMore Atomic Structure Questions
When 3Li7 nuclei are bombarded by protons, and the resultant nuclei are 4Be8, the emitted particles will be...A diatomic molecule is made of two masses m1and m2 which are separated by a distance r. Ifwe calculate its rotational en...The stopping potential double when the frequency of the incident light change from v to . Then the work function of the ...The half life of a radioactive substance against α - decay is 1.2 × 107 s. What is the decay rate for 4.0 ...For the same rise in temperature of one mole of gas at constant volume the heat required for non-linear triatomic gas is...