ThermodynamicsHard
Question
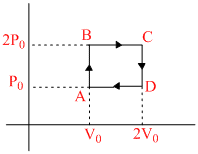
Helium gas goes through a cycle ABCDA(consisting of two isochoric and two isobaric lines) as shown in figure. Efficiency of this cycle is nearly (Assume the gas to be close to ideal gas) :-

Options
A.12.5%
B.15.4%
C.9.1%
D.10.5%
Solution
W = Area bounded by curve = P0V0
QAB = nCvᐃT = n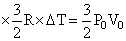
QBC = nCP DT = n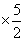 R × ᐃT = 5P0V0
R × ᐃT = 5P0V0
Total heat supplied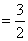 P0V0 + 5P0V0
P0V0 + 5P0V0 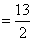 P0V0
P0V0
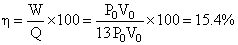
QAB = nCvᐃT = n
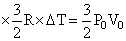
QBC = nCP DT = n
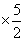 R × ᐃT = 5P0V0
R × ᐃT = 5P0V0Total heat supplied
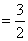 P0V0 + 5P0V0
P0V0 + 5P0V0 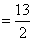 P0V0
P0V0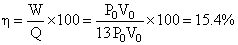
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
In an isometric change :...1 gm of water at 100oC is converted into steam at 100oC. The amount of external work done by the system :...One mole of an ideal gas undergoes a process whose molar heat capacity is 4R and in which work done by gas for small cha...Starting with the same initial conditions, an ideal gas expand from volume V1 to V2 in three different ways, the work do...One mole of a monatomic ideal gas is taken along two cyclic processes E → F → G → E and E → F &#...