ThermodynamicsHard
Question
Starting with the same initial conditions, an ideal gas expand from volume V1 to V2 in three different ways, the work done by the gas is W1 if the process is purely isothermal, W2 if purely isobaric and W3 is purly adiabatic, then
Options
A.W2 > W1 > W3
B.W2 > W3 > W1
C.W1 > W2 > W3
D.W1 > W3 > W2
Solution
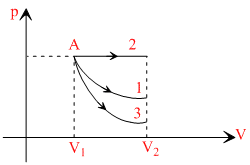
The corresponding p-V graphs (also called indicator diagram) in three different process will be as shown :
Area under the graph gives the work done by the gas
(Area)2 > (Area)1 > (Area)3
∴ W2 > W1 > W3
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
The temperature of an ideal gas is increased from 120 K to 480 K. If at 120 K theroot mean square velocity of the gas mo...A carnot engine working between 300 K and 600 K has work out put of 800 J per cycle. The amount of heat and energy suppl...Cp - Cv = R correctly applicable to which of the following :-...The unit of gas constant R is :-...A refrigerator with coefficient of performance 1/3 releases 200 J of heat to a hot reservoir, then the work done on the ...