ThermodynamicsHard
Question
One mole of an ideal gas undergoes a process whose molar heat capacity is 4R and in which work done by gas for small change in temperature is given by the relation dW = 2RdT, then the ratio 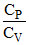 is
is
Options
A.7/5
B.5/3
C.3/2
D.2
Solution
For small change dq = du + dw
nCdT = nCV dT + 2nRdT
∴ C = CV + 2R; 4R = CV + 2R
∴ CV = 2R
Also CV =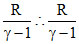 = 2R ⇒ 2γ - 2 = 1 ⇒ γ =
= 2R ⇒ 2γ - 2 = 1 ⇒ γ = 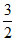
nCdT = nCV dT + 2nRdT
∴ C = CV + 2R; 4R = CV + 2R
∴ CV = 2R
Also CV =
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
10 mole of an ideal gas is undergoing the process shown in the figure. The heat involved in the process from $P_{1}$ to ...When an ideal diatomic gas is heated at constant pressure the fraction of the heat energy supplied which increases the i...On mixing equal volumes of a monoatomic gas and a diatomic gas at the same initial pressure and temprature the ratio of ...A cylinder of radius R made of a material of material of thermal conductivity K2 is surrounded by cylindrical shell of i...An insulated container of gas has two chambers separated by an insulating partition. One of the chambers has volume V1 a...