Chemical BondingHard
Question
The increasing order of the ionic radii of the given isoelectronic species is :-
Options
A.K+, S2-, Ca2+ , Cl-
B.Cl-, Ca2+, K+ , S2-
C.S2-, Cl-, Ca2+, K+
D.Ca2+, K+, Cl-, S2-
Solution
In isoelectronic ions 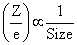
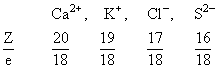
Size Ca2+ < K+ < Cl- < S2-
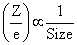
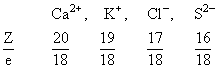
Size Ca2+ < K+ < Cl- < S2-
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In the reaction NH3 + H2O ⇋ NH2- + H3O+ the conjugate base of NH3 is :-...Assuming that Hund′s rule is violated, the bond order and magnetic nature of the diatomic molecule B2 is...Which of the following is a polar molecule ?...Bond order of 1.5 is shown by...Which statements are NOT TRUE about ${XeO}_{2}{\text{ }F}_{2}$ ?A. It has a see-saw shape.B. Xe has 5 electron pairs in ...