Chemical BondingHard
Question
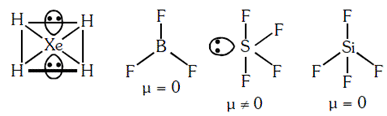
Which of the following is a polar molecule ?
Options
A.XeF4
B.BF3
C.SF4
D.SiF4
Solution
Unsymmetrical distribution of e- cloud leads to the formation of polar molecule

Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
For a given reaction, ∆H = 35.5 kJ mol-1 and ∆S = 83.6 JK-1mol-1. The reaction is spontaneous at : (Assume t...Among the following moleculesThose having same number of lone pair of Xe are :...Bond Angle is maximum in :-...When two equal sized pieces of the same metal at different temperatures Th (hot piece) and Tc(cold piece) are brought in...Which of the two ions from the list given below, have the geometry that is explained by the same hybridisation of orbita...