Chemical BondingHard
Question
Assuming that Hund′s rule is violated, the bond order and magnetic nature of the diatomic molecule B2 is
Options
A.1 and diamagnetic
B.0 and diamagnetic
C.1 and paramagnetic
D.0 and paramagnetic
Solution
B2 (10) = σls2 σ*ls2 σ2s2 σ*2s2 π2px2
Bond order =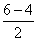 = 1 (nature diamagnetic as no unpaired electron)
= 1 (nature diamagnetic as no unpaired electron)
Bond order =
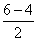 = 1 (nature diamagnetic as no unpaired electron)
= 1 (nature diamagnetic as no unpaired electron)Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
In acidic medium, H2O2 - changes Cr2O7-2 to CrO5 which has two (-O-O-) bonds. Oxidation state of Cr in CrO5 is...In the reacdtion sequence2CH3CHO AB ; the product B is :...The correct stability order for the following species is...Which artificial sweetener contains chlorine ?...Spectrum of Li2+ is similar to that of...