Chemical BondingHard
Question
Bond order of 1.5 is shown by
Options
A.O2+
B.O2-
C.O22-
D.O2
Solution
(a) MO configuration of O2+ ( 8 + 8 - 1 = 15)
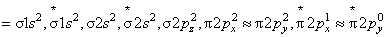
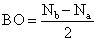
where, Nb = number of electrons in bounding molecular orbital
Na = number of electrons in anti-bounding molecular orbital
∴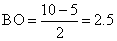
Similarly,
O2- ( 8 + 8 + 1 = 17 )
so BO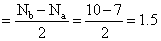
(c)O22- ( 8 + 8 + 2 = 18 )
BO
(d) O2( 8 + 8 = 16 )
BO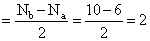
Thus,O2- shows the bond order 1.5.
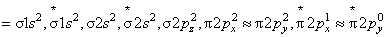
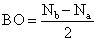
where, Nb = number of electrons in bounding molecular orbital
Na = number of electrons in anti-bounding molecular orbital
∴
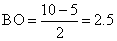
Similarly,
O2- ( 8 + 8 + 1 = 17 )
so BO
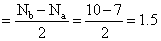
(c)O22- ( 8 + 8 + 2 = 18 )
BO

(d) O2( 8 + 8 = 16 )
BO
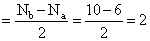
Thus,O2- shows the bond order 1.5.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Inionic part of Cl2O6(s) will be :-...Among of the following, the species having square planar geometry for central for central atom are (i) XeF4 (ii)SF4 (iii...The maximum radius of sphere that can be fitted in the octahedral hole of cubical closed packing of sphere of radius r i...X-X and C-C bond length are respectively 1.0, 1.54(Ao) then C-X bond length would be (ENX = 3.0 ENC = 2.0)...In which of the following ion, the hybrid state of halogen is sp3 :-...