ThermodynamicsHard
Question
The standard Gibbs energy change at 300 K for the reaction 2A ⇋ B + C is 2494.2J. At a given time, the composition of the reaction mixture is [A] = 1/2, [B] = 2 and [C] = 1/2. The reaction proceeds in the : [R = 8.314 J/K/mol, e = 2.718]
Options
A.forward direction because Q < Kc
B.reverse direction because Q < Kc
C.forward direction because Q > Kc
D.reverse direction because Q > Kc
Solution
ᐃGo = –RT ln Keq
⇒ 2494.2 = −8.314 × 300 ln Keq
⇒ Keq =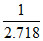
Now for reaction ⇒ 2A ⇒ B + C
Q =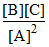
=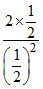 = 4
= 4
∴ Q > Kc, hence reverse direction
⇒ 2494.2 = −8.314 × 300 ln Keq
⇒ Keq =
Now for reaction ⇒ 2A ⇒ B + C
Q =
=
∴ Q > Kc, hence reverse direction
Create a free account to view solution
View Solution FreeMore Thermodynamics Questions
With what minimum pressure must a given volume of an ideal gas ($\gamma$ = 1.4), originally at 400 K and 100 kPa, be adi...The standard molar entropy of an ideal gas $\left( \gamma = \frac{4}{3} \right)$is 2.5 cal/K-mol at 25°C and 1 bar. The ...A reversible heat engine absorbs 40 kJ of heat at 500 K and performs 10 kJ of work rejecting the remaining amount to the...The minimum work which must be done to compress 16 g of oxygen isothermally at 300 K from a pressure of 1.01325 × 103 N/...One mole of an ideal gas at 300 K is expended isothermally from an initial volume of 1 liter to 10 liters. The value of ...