General Organic ChemistryHard
Question
In Carius method of estimation of halogens, 250 mg of an organic compound gave 141 mg of AgBr. The percentage of bromine in the compound is : (at. mass Ag = 108; Br = 80)
Options
A.48
B.60
C.24
D.36
Solution
Organic compound 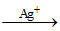 AgBr
AgBr
=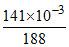
Number of moles of AgBr = Number of moles of Br- =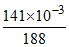
∴ mass of Br =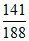 × 10−3 × 80
× 10−3 × 80
∴ mass % of Br =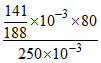 × 100
× 100
= 24
=
Number of moles of AgBr = Number of moles of Br- =
∴ mass of Br =
∴ mass % of Br =
= 24
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Which of the following compound will exhibit geomrtrical isomerism ?...The strongest acid amongst the following compounds is :...The hybridisation of carbon atoms in C - C single bond of H - C ≡ C - CH = CH2 is :...Given the cyclohexanol (I), acetic acid (II), 2, 4, 6 -trinitrophenol (III) and phenol (IV). In these, the order of decr...In each of the following pairs of species which species is an electrophile :...