General Organic ChemistryHard
Question
The strongest acid amongst the following compounds is :
Options
A.HCOOH
B.CH3 CH2 CH (Cl) CO2H
C.CICH2 CH2 CH2 COOH
D.CH3 COOH
Solution
Electron releasing groups (Alkyl groups) de stabilizes conjugate base. The +I effect of
C3 H7 is less than - I effect of Cl
Ka of HCOOH is 17.9 × 10-5
Ka of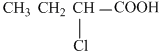 is 139 × 10-5
is 139 × 10-5
C3 H7 is less than - I effect of Cl
Ka of HCOOH is 17.9 × 10-5
Ka of
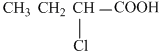 is 139 × 10-5
is 139 × 10-5 Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Keto-enol tautomerism is observed in :...Which of the following exhibit electromeric effect -...Which of the following belongs to + I group...Arrange pH of the given compounds in decreasing order: (1) Phenol (2) Ethyl alcohol (3) Formic acid (4) Benzoic acid...The correct acidity order of the following is...