General Organic ChemistryHard
Question
The hybridisation of carbon atoms in C - C single bond of H - C ≡ C - CH = CH2 is :
Options
A.sp3 - sp3
B.sp2 - sp3
C.sp - sp2
D.sp3 - sp
Solution
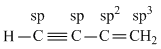
Thus C - C single bond is sp - sp2 σ - bond.
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
Rank the following free radicals in order of decreasing stability : (I) C6H5 HC6H5(II) C6H5 − H − CH = CH2(I...Electrophile O2 attacks the following I II III IVIn which cases cases will be meta-position :...Heterolytic fission of carbon-chlorine bond produces :...The correct statement regarding electrophile is :-...Acetate ion contains :...