General Organic ChemistryHard
Question
Given the cyclohexanol (I), acetic acid (II), 2, 4, 6 -trinitrophenol (III) and phenol (IV). In these, the order of decreasing acidic character will be
Options
A.III > II > IV > I
B.II > III > I > IV
C.II > III > IV > I
D.III > IV > II > I
Solution
Higher the tendency to give a proton, higher the acidic character, and tendency to lose a proton depends upon the stability of intermediate, ie, carbanion formed.
2, 4, 6-trinitrophenol after the loss of as proton gives 2,4,6- rinitrophenoxide ion which is stabilised by resonance, -I effect and -M effect, thus is most acidic among the given compounds.
Phenol after losing a proton form phenoxide ion which is also stabilised by resonance, -M and -I effect but it less stabilised as compared to 2, 4, 6-trinitrophenoxide ions Thus it is less acidic as compared to 2 ,4 ,6-trinitrophenol.
(CH3COOH) after losing a proton gives acetate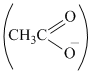 ion which is stabilised by only resonance. However, it is more resonance stabilised as compared to phenoxide ion, thus more acidic as compared to a phenol. 2, 4, 6- trinitrophenol, however , is more acetic than acetic acid due to the presence of three electron withdrawing NO2 groups. Cyclohexanol gives an anion that is least stable among the given, thus, it is least acidic,
ion which is stabilised by only resonance. However, it is more resonance stabilised as compared to phenoxide ion, thus more acidic as compared to a phenol. 2, 4, 6- trinitrophenol, however , is more acetic than acetic acid due to the presence of three electron withdrawing NO2 groups. Cyclohexanol gives an anion that is least stable among the given, thus, it is least acidic,
Hence, the correct order of acidic strength is 2,4,6-trinitrophenol > acetic acid > phenol > cyclohexanol
III > II > IV > I
2, 4, 6-trinitrophenol after the loss of as proton gives 2,4,6- rinitrophenoxide ion which is stabilised by resonance, -I effect and -M effect, thus is most acidic among the given compounds.
Phenol after losing a proton form phenoxide ion which is also stabilised by resonance, -M and -I effect but it less stabilised as compared to 2, 4, 6-trinitrophenoxide ions Thus it is less acidic as compared to 2 ,4 ,6-trinitrophenol.
(CH3COOH) after losing a proton gives acetate
 ion which is stabilised by only resonance. However, it is more resonance stabilised as compared to phenoxide ion, thus more acidic as compared to a phenol. 2, 4, 6- trinitrophenol, however , is more acetic than acetic acid due to the presence of three electron withdrawing NO2 groups. Cyclohexanol gives an anion that is least stable among the given, thus, it is least acidic,
ion which is stabilised by only resonance. However, it is more resonance stabilised as compared to phenoxide ion, thus more acidic as compared to a phenol. 2, 4, 6- trinitrophenol, however , is more acetic than acetic acid due to the presence of three electron withdrawing NO2 groups. Cyclohexanol gives an anion that is least stable among the given, thus, it is least acidic, Hence, the correct order of acidic strength is 2,4,6-trinitrophenol > acetic acid > phenol > cyclohexanol
III > II > IV > I
Create a free account to view solution
View Solution FreeMore General Organic Chemistry Questions
The synthesis of alkyl fluoride is best accomplished by :...Give the correct order of decreasing basicity of the following compounds,C6H5NH2 (C6H5)2NH (C6H5)3N C6H11NH2 I II III IV...The number of sigma and pi-bonds in 1-butene-3-yne are :...Among the following which one have a meso form ?...I II IIIArrange following phenol in increasing order of Pka value...