Chemical BondingHard
Question
The intermolecular interaction that is dependent on the inverse cube of distance between the molecules is :-
Options
A.London force
B.Hydrogen bond
C.ion-ion interaction
D.ion-dipol interaction
Solution
The intermolecular interaction that is dependent on 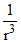 (where r is the distance between the molecules). When intermolecular interaction is considered in terms of energy then, it is true for dipoledipole intereaction and the hydrogen bond is the special kind of dipole-dipole intereaction. Hence the answer is 2. If the intermolecular interaction is considered in terms of force then, it is true for ion-dipole intereaction and answer will be 4.
(where r is the distance between the molecules). When intermolecular interaction is considered in terms of energy then, it is true for dipoledipole intereaction and the hydrogen bond is the special kind of dipole-dipole intereaction. Hence the answer is 2. If the intermolecular interaction is considered in terms of force then, it is true for ion-dipole intereaction and answer will be 4.
Create a free account to view solution
View Solution FreeMore Chemical Bonding Questions
Among the following oxoacids, the correct decreasing order of acid strength is...Gas Liberated during reaction :-...Which of the following statements is/are wrong ?The following reactions occur:i. P4 + 5O2 → P4O10.ii. P4 + 3O2 ...The product formed in the reaction of SOCl2 with white phosphorous is...If at 298 K the bond energies of C-H, C-C, C = C and H-H bonds are respectively 414, 347, 615 and 435 kJ mol-1, the valu...